Simultaneous optimization of factors affecting native starch pretreatment and enzymatic hydrolysis using magnetic covalent immobilized α-amylase
Keywords:
Native starch, , Thermal hydrolysis , Sonication treatment , Immobilized enzyme , Magnetic nanoparticlesAbstract
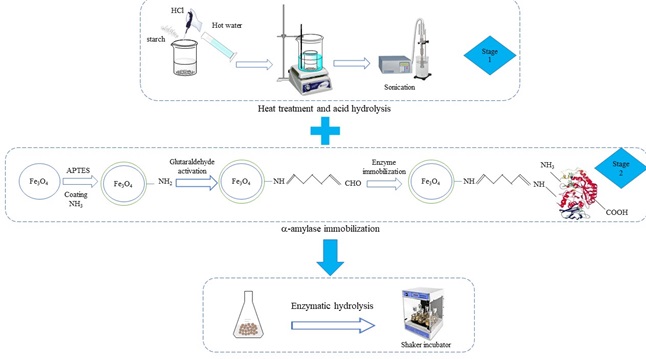
In this study, for the first time the effects of different factors on hydrolysis of low concentration of corn starch were investigated in two steps. In the first step, thermal hydrolysis with pH variation and sonication treatment and finally enzymatic hydrolysis of starch were investigated by one factor at a time method. The best conditions of maximum reducing sugar production and maximum releasing of amylose were starch with pH=4.5, sonication time 30 min and 24 hours solution storage in 4 °C with enzymatic hydrolysis by 335 ppm immobilized enzyme for 15 min. In the second step, the effects of three variables on starch hydrolysis to determine the optimal conditions were investigated by 23 factorial design with multiple responses desirability method. Starch concentration 0.5% (w/v), initial starch pH of 4.5 and enzymatic hydrolysis temperature 50°C were determined as optimum conditions for achieving maximum changes in reducing sugar concentration 92.48 (mg/g starch) and ratio of final and initial soluble amylose content of hydrolysate solution 0.62 (by more than 75% separation of immobilized enzyme on MNPs after the process by external magnetic field. Also, experiment showed the possibility of immobilized enzyme reusing, which can retain 40% of its ability to produce reducing sugar and retain magnetic features of nanoparticles after 6 cycles.
Downloads
References
[1] Guo, H., Tang, Y., Yu, Y., Xue, L., Qian, J. q., Covalent immobilization of α-amylase on magnetic particles as catalyst for hydrolysis of high-amylose starch. International journal of biological macromolecules, 2016. 87: p. 537-544. http://doi.org/10.1016/j.ijbiomac.2016.02.080
[2] Wang, S. and Copeland, L., Effect of Acid Hydrolysis on Starch Structure and Functionality: A Review. Critical Reviews in Food Science and Nutrition, 2015. 55(8): p. 1079-1095. http://doi.org/10.1080/10408398.2012.684551
[3] Ahmed, N.E., El Shamy, A.R., Awad, H.M., Optimization and immobilization of amylase produced by Aspergillus terreus using pomegranate peel waste. Bulletin of the National Research Centre, 2020. 44(1): p. 1-12. doi: https://doi.org/10.1186/s42269-020-00363-3
[4] Cao, L., Carrier-bound Immobilized Enzymes: Principles, Applications and Design, John Wiley & sons, 2006. DOI:10.1002/3527607668
[5] Li, Z., Liu, W., Gu, Z., Li, C., Hong, Y., Cheng, L., The effect of starch concentration on the gelatinization and liquefaction of corn starch. Food Hydrocolloids, 2015. 48: p. 189-196. https://doi.org/10.1016/j.foodhyd.2015.02.030
[6] Din, Z. u., Xiong, H., Fei, P., Physical and Chemical Modification of Starches - A Review. Critical reviews in food science and nutrition, 2015. 57. http://doi.org/10.1080/10408398.2015.1087379
[7] Sumarno, Bramantyo, A., Febriyati, P., Gunardi, I., Trisanti, P. N., Ultrasound Pre-treatment for Intensification of Hydrothermal Process in Reducing Sugar Production from Cassava Starch. IOP Conference Series: Materials Science and Engineering, 2019. 543(1): p. 012085. http://doi.org/10.1088/1757-899X/543/1/012085
[8] Maniglia, B.C., Castanha, N., Le-Bail, P., Le-Bail, A., Augusto, P. E. D., Starch modification through environmentally friendly alternatives: a review. Critical Reviews in Food Science and Nutrition, 2021. 61(15): p. 2482-2505. http://doi.org/10.1080/10408398.2020.1778633
[9] Kaur, B., Ariffin, F., Bhat, R., Karim, A. A., Progress in starch modification in the last decade. Food Hydrocolloids, 2012. 26(2): p. 398-404. https://doi.org/10.1016/j.foodhyd.2011.02.016
[10] Tester, R. F. and Debon, S.J.J., Annealing of starch — a review. International Journal of Biological Macromolecules, 2000. 27(1): p. 1-12. http://doi.org/10.1016/s0141-8130(99)00121-x
[11] Uthumporn, U., Zaidul, I. S. M., Karim, A. A., Hydrolysis of granular starch at sub-gelatinization temperature using a mixture of amylolytic enzymes. Food and Bioproducts Processing, 2010. 88(1): p. 47-54. https://doi.org/10.1016/j.fbp.2009.10.001
[12] Falsafi, S. R., Maghsoudlou, Y., Rostamabadi, H., Rostamabadi, M. M., Hamedi, H., Hosseini, S. M. H., Preparation of physically modified oat starch with different sonication treatments. Food Hydrocolloids, 2019. 89: p. 311-320. http://doi.org/10.1016/j.foodhyd.2018.10.046
[13] Iida, Y., Tuziuti, T., Yasui, K., Towata, A., Kozuka, T., Control of viscosity in starch and polysaccharide solutions with ultrasound after gelatinization. Innovative Food Science & Emerging Technologies, 2008. 9(2): p. 140-146. https://doi.org/10.1016/j.ifset.2007.03.029
[14] Wang, D., et al., Ultrasound assisted enzymatic hydrolysis of starch catalyzed by glucoamylase: Investigation on starch properties and degradation kinetics. Carbohydrate polymers, 2017. 175: p. 47-54. https://doi.org/10.1016/j.carbpol.2017.06.093
[15] Wang, D., et al., Study on the mechanism of ultrasound-accelerated enzymatic hydrolysis of starch: Analysis of ultrasound effect on different objects. International Journal of Biological Macromolecules, 2020. 148: p. 493-500. https://doi.org/10.1016/j.ijbiomac.2020.01.064
[16] Arfin, T. and Tarannum, A., Chapter 6 - Engineered Nanomaterials for Industrial Application: An Overview, in Handbook of Nanomaterials for Industrial Applications, C. Mustansar Hussain, Editor. 2018, Elsevier. p. 127-134. https://doi.org/10.1016/B978-0-12-813351-4.00006-7
[17] Wee, L.L., Annuar, M., Ibrahim, S., Chisti, Y., Enzyme-mediated production of sugars from sago starch: statistical process optimization. Chemical Engineering Communications, 2011. 198(11): p. 1339-1353. https://doi.org/10.1080/00986445.2011.560513
[18] Eslamipour, F. and Hejazi, P., Effects of surface modification and activation of magnetic nanoparticles on the formation of amylase immobilization bonds under different ionic strength conditions. Journal of Molecular Catalysis B: Enzymatic, 2015. 119: p. 1-11. https://doi.org/10.1016/j.molcatb.2015.05.006
[19] Sirohi, R., et al., Acid hydrolysis of damaged wheat grains: Modeling the formation of reducing sugars by a neural network approach. Industrial Crops and Products, 2020. 149: p. 112351. https://doi.org/10.1016/j.indcrop.2020.112351
[20] Bradford, M.M., A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry, 1976. 72(1): p. 248-254. https://doi.org/10.1016/0003-2697 (76)90527-3
[21] Miller, G.L., Use of Dinitrosalicylic Acid Reagent for Determination of Reducing Sugar. Analytical Chemistry, 1959. 31(3): p. 426-428. https://doi.org/10.1021/ac60147a030
[22] McGrance, S.J., Cornell, H. J., Rix, C. J, .A Simple and Rapid Colorimetric Method for the Determination of Amylose in Starch Products. Starch - Stärke, 1998. 50(4): p. 158-163. https://doi.org/10.1002/(SICI)1521-379X(199804)50:43.0.CO;2-7
[23] Zhang, Y., et al., Ultrasound-assisted preparation of octenyl succinic anhydride modified starch and its influence mechanism on the quality. Food Chemistry: X, 2020. 5: p. 100077. https://doi.org/10.1016/j.fochx.2020.100077
[24] Chan, H. T., Bhat, R. Karim, A. A., Effects of sodium dodecyl sulphate and sonication treatment on physicochemical properties of starch. Food Chemistry, 2010. 120(3): p. 703-709. https://doi.org/10.1016/j.foodchem.2009.10.066
[25] Zhang, H., Hou, H., Liu, P., Wang, W., Dong, H., Effects of acid hydrolysis on the physicochemical properties of pea starch and its film forming capacity. Food Hydrocolloids, 2019. 87: p. 173-179. https://doi.org/10.1016/j.foodhyd.2018.08.009
[26] Aparicio-Saguilán, A., Flores-Huicochea, E., Tovar, J., García-Suárez, F., Gutiérrez-Meraz, F., Bello-Pérez, L., Resistant Starch-rich Powders Prepared by Autoclaving of Native and Lintnerized Banana Starch: Partial Characterization. Starch - Stärke, 2005. 57(9): p. 405-412. https://doi.org/10.1002/star.200400386
[27] Koksel, H., Ozturk, S., Kahraman, K., Basman, A., Ozbas, O. O., Ryu, G. H., Evaluation of molecular weight distribution, pasting and functional properties, and enzyme resistant starch content of acid-modified corn starches. Food Science and Biotechnology, 2008. 17(4): p. 755-760.
[28] Nasrin, T. A. A. and Anal, A. K., Resistant starch III from culled banana and its functional properties in fish oil emulsion. Food Hydrocolloids $V 35, 2014: p. 403-409.
https://doi.org/10.1016/j.foodhyd.2013.06.019
[29] Li, Z., Cai, L., Gu, Z., Shi, Y. C., Effects of Granule Swelling on Starch Saccharification by Granular Starch Hydrolyzing Enzyme. Journal of Agricultural and Food Chemistry, 2014. 62(32): p. 8114-8119. https://doi.org/10.1021/jf500814g
[30] Bej, B.,. Basu, R.K, Ash, S. N., Kinetic studies on acid catalysed hydrolysis of starch. Journal of Scientific and Industrial Research, 2008. 67(4): p. 295-298.
[31] Tian, J., et al., Existing cell wall fragments modify the thermal properties and hydrolysis of potato starch. Food Hydrocolloids, 2018. 85: p. 229-232. https://doi.org/10.1016/j.foodhyd.2018.07.033
[32] Brodnjak, U.V., Improvement of physical and optical properties of chitosan-rice starch films pre-treated with ultrasound. Bulg. Chem. Commun., 2017. 49: p. 859-867.
[33] Pal, S., Mal, D., Singh, R., Cationic Starch: An effective flocculating agent. Carbohydrate Polymers, 2005. 59: p. 417-423. https://doi.org/10.1016/j.carbpol.2004.06.047
[34] Azmi, A., Malek, M., Puad, N., A review on acid and enzymatic hydrolyses of sago starch. International Food Research Journal, 2017. 24(12): p. 265-273.
[35] Gangadharan, D., Madhavan Nampoothiri, K., Sivaramakrishnan, S., Pandey, A., Immobilized bacterial α-amylase for effective hydrolysis of raw and soluble starch. Food Research International, 2009. 42(4): p. 436-442. https://doi.org/10.1016/j.foodres.2009.02.008
[36] Tüzmen, N., Kalburcu, T., Denizli, A., α-Amylase immobilization onto dye attached magnetic beads: Optimization and characterization. Journal of Molecular Catalysis B: Enzymatic, 2012. 78: p. 16-23. https://doi.org/10.1016/j.molcatb.2012.01.017
[37] Dhavale, R., et al., α-amylase immobilized on magnetic nanoparticles: reusable robust nano-biocatalyst for starch hydrolysis. Materials Research Express, 2018. 5(7): p. 075403. http://doi.org/ 10.1088/2053-1591/aacef1
Downloads
Published
Submitted
Revised
Accepted
Issue
Section
License
Copyright (c) 2025 Shadi Khazaei (Author); Parisa Hejazi

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.